Latest News on Your Doctor's Blog
Loading…
Rabeprazole (EC) 40mg + Domperidone (SR) 30mg Capsules Manufacturer, Supplier & Exporter in India Manufacturer of Rabeprazole + Domperidone Capsules in India We are a reputed and globally recognized pharmaceutical manufacturer engaged in the production, supply, and export of high-quality gastroenterology formulations. Among our key offerings, Rabeprazole (EC) 40mg + Domperidone (SR) 30mg Capsules stand as a trusted therapeutic combination widely used in the treatment of acid-related disorders. Manufactured under stringent quality standards, our formulation is designed to deliver effective relief in conditions such as gastroesophageal reflux disease (GERD), acid reflux, heartburn, and peptic ulcer disease. With advanced production infrastructure and WHO-GMP compliant facilities, we ensure consistent purity, safety, and efficacy in every capsule. About Rabeprazole + Domperidone Capsules Composition Rabeprazole 40mg Domperidone 30mg Capsules This combination consists of two well-established pharmaceutical agents: Rabeprazole (Enteric Coated 40mg) – A proton pump inhibitor (PPI) Domperidone (Sustained Release 30mg) – A prokinetic agent Together, these components provide dual-action therapy by controlling excess stomach acid production while improving gastrointestinal motility. This synergy makes it highly effective for patients suffering from chronic acidity and related digestive discomfort. Mechanism of Action Rabeprazole – Acid Suppression Therapy Rabeprazole works by blocking the hydrogen-potassium ATPase enzyme system […]
Epalrestat Sustained Release Tablets 150mg Manufacturer, Supplier & Exporter in India Leading Epalrestat 150mg Sustained Release Tablets Manufacturer in India We at Taj Medico are a well-established manufacturer, exporter, and supplier of Epalrestat Sustained Release Tablets 150mg in India, delivering consistent quality, purity, and pharmaceutical excellence. Our production facility is based in Vapi, Gujarat, and is equipped with advanced manufacturing infrastructure designed to meet stringent international quality standards. Epalrestat Tablets 150mg are widely used in the management of diabetic neuropathy, a complication of long-standing or poorly controlled diabetes. We ensure that every batch of tablets is manufactured under strict quality control procedures to maintain safety, efficacy, and stability. Our commitment extends beyond manufacturing—we focus on global healthcare support by supplying high-quality Epalrestat SR Tablets to Africa, the Middle East, Latin America (LTAM), and other regulated and semi-regulated markets. What is Epalrestat Sustained Release 150mg? Epalrestat Sustained Release Tablets 150mg Epalrestat 150mg Sustained Release Tablets belong to a class of medicines known as aldose reductase inhibitors. This medicine is specifically designed to help manage diabetic neuropathy, a condition where prolonged high blood sugar levels damage peripheral nerves. Diabetic neuropathy commonly results in symptoms such as: Burning sensation in limbs Tingling and […]
Orlistat Capsules 120mg Manufacturer Supplier in India Orlistat Capsules 120mg Manufacturer in India We are a trusted Orlistat Capsules 120mg manufacturer in India, delivering premium-quality formulations for domestic and international pharmaceutical markets. Our manufacturing facility in Vapi, Gujarat is equipped with advanced production technology, strict quality systems, and skilled pharmaceutical professionals committed to producing safe and effective anti-obesity medicines. Orlistat Capsules 120mg is widely prescribed for weight management in obese and overweight individuals. It works by reducing the absorption of dietary fat in the intestine, helping patients control calorie intake and support long-term weight reduction when combined with a balanced diet and exercise plan. As a reliable manufacturer and exporter, we supply Orlistat Capsules 120mg in bulk quantities, private label packaging, contract manufacturing models, and PCD pharma franchise opportunities across India and overseas markets. What is Orlistat Capsules 120mg? Orlistat Capsules 120mg Orlistat 120mg is a clinically recognized lipase inhibitor used for the treatment of obesity and weight-related metabolic disorders. It acts locally in the stomach and small intestine by blocking enzymes that break down fats from food. As a result, a portion of consumed fat remains undigested and is naturally eliminated from the body. This medicine is commonly prescribed […]
Potassium Iodate Tablets 170mg Manufacturer Supplier in India Potassium Iodate Tablets 170mg Manufacturer in India We are recognized among the leading Potassium Iodate Tablets 170mg manufacturers in India, delivering premium quality pharmaceutical formulations with strict quality standards, consistent potency, and reliable supply capabilities. Our advanced manufacturing facilities follow international GMP guidelines, ensuring every batch meets required pharmaceutical specifications for domestic and export markets. Potassium Iodate Tablets 170mg are widely used as a thyroid blocking agent during radiation emergencies and for specific medical indications where iodine supplementation or thyroid protection is required under professional supervision. We manufacture and supply these tablets with precision, ensuring stability, purity, and compliance with global pharmaceutical norms. Our commitment to excellence, ethical business practices, and customer satisfaction has positioned us among the most dependable names for Potassium Iodate Tablets 170mg supplier and exporter in India. What is Potassium Iodate Tablets 170mg? Potassium Iodate Tablets 170mg Potassium Iodate Tablets 170mg contain a stable iodine compound designed for controlled pharmaceutical use. This medicine is commonly used to help protect the thyroid gland from absorbing radioactive iodine during nuclear emergencies when advised by authorities. It may also be utilized in specific healthcare applications based on medical recommendation. The thyroid […]
Orlistat Capsules 60mg Manufacturer Supplier in India Orlistat Capsules 60mg Manufacturer in India We are a trusted Orlistat Capsules 60mg manufacturer in India, delivering premium-quality pharmaceutical formulations for domestic and international markets. With advanced manufacturing capabilities, strict quality systems, and a customer-focused approach, we supply Orlistat Capsules 60mg to distributors, wholesalers, hospitals, pharmacies, and healthcare institutions worldwide. Our manufacturing unit in Vapi, Gujarat, India follows stringent GMP standards to ensure every batch meets high levels of purity, safety, stability, and consistency. We specialize in third-party manufacturing, private labeling, contract manufacturing, and global exports of Orlistat Capsules 60mg. What is Orlistat Capsules 60mg? Orlistat Capsules 60mg (Fetrasil) Orlistat Capsules 60mg is widely used for the management of obesity and weight reduction in overweight individuals. It is recommended along with a calorie-controlled diet, physical activity, and healthy lifestyle changes. Orlistat works by reducing the absorption of dietary fats in the body. This medicine acts locally in the digestive tract and supports long-term weight management when used under medical supervision. How Orlistat Capsules 60mg Works Orlistat belongs to a class of medicines called lipase inhibitors. It blocks the enzymes in the stomach and small intestine responsible for breaking down fats from food. As […]
Clozapine 100mg Tablets Manufacturer in India Leading Clozapine 100mg Tablets Manufacturer in India We are recognized as a trusted Clozapine 100mg Tablets manufacturer in India, delivering high-quality pharmaceutical formulations with assured purity, safety, and consistent therapeutic effectiveness. At Taj Medico, we specialize in manufacturing and supplying premium-grade Clozapine 100mg Tablets for domestic and international markets with strong regulatory compliance and advanced production standards. Our manufacturing unit based in Vapi, Gujarat, India, operates with modern pharmaceutical infrastructure, ensuring reliable bulk production, timely dispatch, and customized packaging solutions for distributors, hospitals, importers, wholesalers, and PCD franchise partners. About Clozapine 100mg Tablets Clozapine 100mg Tablets Clozapine 100mg Tablets contain Clozapine, an atypical antipsychotic medicine widely prescribed for the management of severe schizophrenia and treatment-resistant psychiatric disorders. It is commonly used when patients do not respond adequately to conventional antipsychotic therapies. This medicine works by balancing neurotransmitters in the brain that influence mood, perception, behavior, and thought processes. It is particularly beneficial in reducing hallucinations, delusions, emotional withdrawal, and recurrent suicidal tendencies associated with schizophrenia. Therapeutic Uses of Clozapine 100mg Tablets We supply Clozapine 100mg Tablets for multiple psychiatric treatment requirements, including: Treatment-resistant schizophrenia Severe psychotic disorders Schizoaffective disorders Recurrent suicidal behavior in schizophrenia […]
Clozapine 25mg Tablets Manufacturer Supplier in India Clozapine 25mg Tablets by Taj Medico – Quality Manufacturing for Global Markets We are recognized as a dependable Clozapine 25mg Tablets manufacturer in India, delivering high-quality psychiatric medicines that meet international standards. Taj Medico has built a strong reputation for supplying premium pharmaceutical formulations to domestic and overseas markets with consistency, safety, and timely service. Our manufacturing operations are based in Vapi, Gujarat, India, one of the leading pharmaceutical hubs known for advanced production infrastructure and regulatory compliance. We manufacture Clozapine 25mg Tablets with a strong focus on purity, efficacy, and reliable packaging for hospitals, distributors, pharmacies, government tenders, and healthcare institutions. Clozapine is a widely prescribed atypical antipsychotic medicine used in the management of schizophrenia, especially treatment-resistant schizophrenia. It is also used in reducing suicidal behavior in certain psychiatric conditions under medical supervision. Clozapine 25mg Tablets Manufacturer in India Clozapine 25mg We are committed to producing Clozapine 25mg Tablets using high-grade active pharmaceutical ingredients and controlled manufacturing processes. Every stage of production is monitored by experienced quality teams to ensure uniformity in every batch. Our facilities follow current Good Manufacturing Practices, ensuring: Accurate formulation and dosage strength Stable shelf life Hygienic production […]
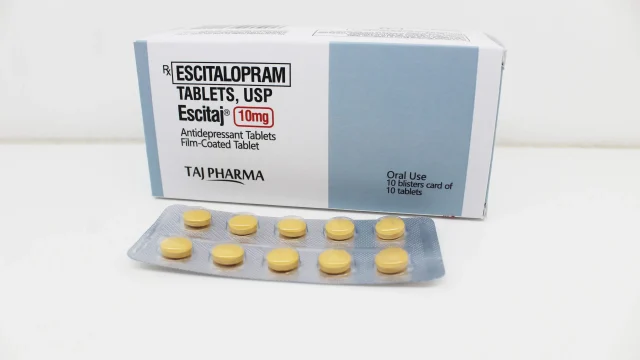
Escitalopram 5mg Tablets Manufacturer in India Escitalopram 5mg Tablets Manufacturer in India We are recognized as a trusted Escitalopram 5mg Tablets Manufacturer in India, offering premium quality formulations manufactured under strict pharmaceutical standards. Our Escitalopram 5mg tablets are widely prescribed for the treatment of Major Depressive Disorder (MDD) and Generalized Anxiety Disorder (GAD) in adults and adolescents. With advanced manufacturing facilities, regulatory compliance, and a commitment to product consistency, we supply Escitalopram tablets across India and international markets. Escitalopram is one of the most widely used selective serotonin reuptake inhibitors (SSRIs), known for improving mood balance, reducing anxiety symptoms, and enhancing emotional well-being. We ensure that every batch of Escitalopram 5mg tablets meets global quality benchmarks for safety, purity, and efficacy. Leading Escitalopram 5mg Tablets Supplier in India Escitalopram 5mg Tablets We are a dependable Escitalopram 5mg Tablets Supplier in India, serving hospitals, wholesalers, pharmacies, healthcare institutions, distributors, and government tenders. Our supply chain is designed for timely delivery, secure packaging, and uninterrupted availability. Key Supply Advantages: PAN India distribution network Bulk order fulfillment capacity Secure blister and strip packaging Competitive manufacturer pricing Fast dispatch and reliable logistics Custom labeling and third-party manufacturing options We maintain a strong reputation for […]
Escitalopram 10mg Tablets Manufacturer in India Leading Escitalopram 10mg Tablets Manufacturer and Exporter in India We are recognized as a trusted Escitalopram 10mg Tablets Manufacturer in India, delivering premium pharmaceutical formulations with high standards of quality, purity, and global compliance. At Taj Medico, we specialize in manufacturing and exporting Escitalopram Tablets for domestic and international markets including Africa, Middle East, LATAM, Asia, and CIS countries. Our advanced manufacturing facilities, stringent quality systems, and commitment to healthcare excellence have made us a preferred source for buyers seeking reliable antidepressant medicines at competitive prices. Escitalopram 10mg Tablets are widely prescribed for the treatment of major depressive disorder (MDD) and generalized anxiety disorder (GAD). These tablets belong to the Selective Serotonin Reuptake Inhibitor (SSRI) class and help improve serotonin levels in the brain to stabilize mood, reduce anxiety, and restore emotional balance. What is Escitalopram 10mg Tablet? Escitalopram 10mg Tablets Escitalopram is an antidepressant medicine used to manage several mental health conditions. It works by increasing serotonin activity in the brain, helping patients feel calmer, emotionally balanced, and more focused. Common Uses of Escitalopram 10mg Tablets Major Depressive Disorder Generalized Anxiety Disorder Panic Disorders Social Anxiety Disorder Obsessive Thoughts (as prescribed) Emotional Stress […]
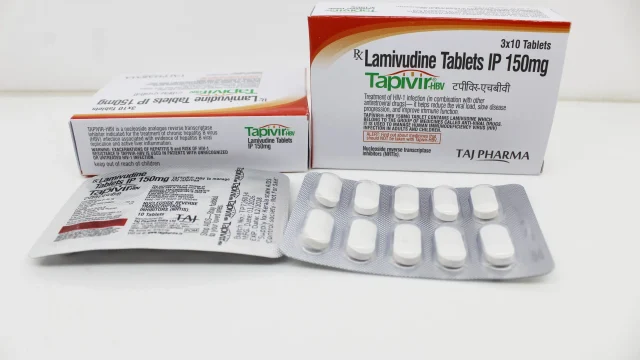
Lamivudine Tablets 150mg Manufacturer in India We at Taj Medico are a trusted and well-established manufacturer, exporter, and supplier of Lamivudine Tablets 150mg (Tapivir) in India. Based in Vapi, Gujarat, we are committed to delivering pharmaceutical products that meet stringent international quality standards, ensuring safety, efficacy, and reliability for global healthcare needs. Lamivudine is a widely prescribed antiviral medication used in the management of HIV infection and chronic Hepatitis B virus (HBV) infection. As a nucleoside reverse transcriptase inhibitor (NRTI), it plays a vital role in antiretroviral therapy (ART) by reducing viral replication and improving immune function. About Lamivudine Tablets 150mg (Tapivir) – Composition & Therapeutic Use Lamivudine Tablets 150 mg Lamivudine Tablets 150mg (Tapivir) are formulated to inhibit the reverse transcriptase enzyme, which is essential for viral replication in HIV and HBV infections. By blocking this process, Lamivudine helps reduce viral load and slows disease progression. Key Therapeutic Applications: Treatment of HIV-1 infection Management of chronic Hepatitis B virus (HBV) infection Used in combination therapy for ART regimens Helps reduce risk of liver damage in HBV patients Improves immune system response in HIV patients Lamivudine is never used alone for HIV treatment; it is always administered as part of […]
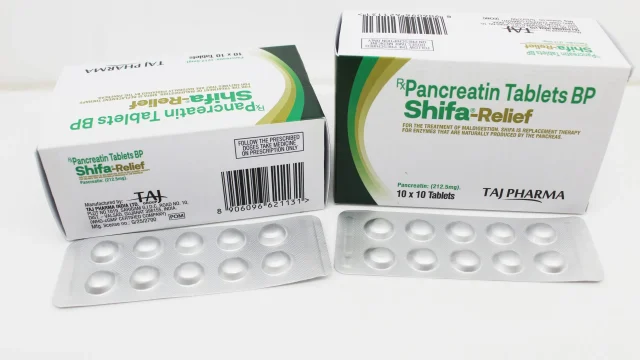
Pancreatin BP 212.5mg Tablets Manufacturer, Supplier and Exporter in India Leading Manufacturer of Pancreatin BP 212.5mg Tablets in India We at Taj Medico are a reputed and well-established manufacturer, exporter, and supplier of Pancreatin BP 212.5mg Tablets in India. Based in Vapi, Gujarat, we are committed to delivering high-quality pharmaceutical formulations that meet stringent international quality standards. Our Pancreatin tablets are formulated with precision to ensure maximum efficacy, safety, and consistency for patients suffering from pancreatic enzyme deficiencies. As a trusted pharmaceutical company, we focus on innovation, quality assurance, and global distribution capabilities, making us a preferred choice for Pancreatin BP 212.5mg Tablets Manufacturer in India, Africa, Middle East, and Latin America (LATAM) markets. What is Pancreatin BP 212.5mg and Its Therapeutic Importance Pancreatin BP 212.5mg Tablets Pancreatin BP 212.5mg Tablets contain a mixture of digestive enzymes including amylase, lipase, and protease, which play a vital role in the digestion of carbohydrates, fats, and proteins. This formulation is primarily used in the treatment of: Pancreatic enzyme deficiency Chronic pancreatitis Cystic fibrosis-related digestive disorders Post-surgical pancreatic insufficiency By supplementing natural digestive enzymes, Pancreatin helps improve nutrient absorption and reduces symptoms such as bloating, abdominal discomfort, and fatty stools. High-Quality Manufacturing Standards […]
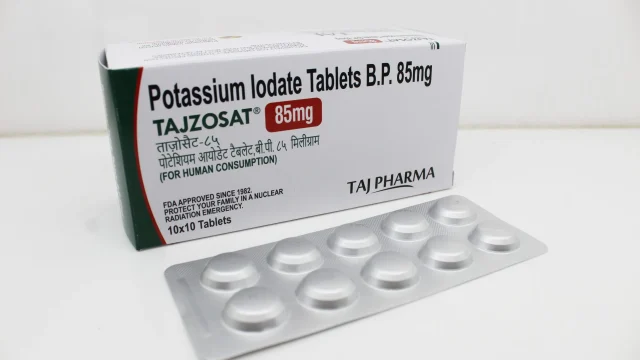
Potassium Iodate Tablets 85mg Manufacturer in India Potassium Iodate Tablets 85mg Manufacturer, Exporter & Supplier in India We at Taj Medico are a trusted and well-established Potassium Iodate Tablets 85mg manufacturer in India, delivering high-quality pharmaceutical formulations that meet global standards of safety, purity, and efficacy. With advanced infrastructure located in Vapi, Gujarat, India, we specialize in the manufacturing, export, and supply of Potassium Iodate Tablets 85mg (TAJZOSAT) for domestic and international markets. Our commitment to excellence has positioned us as a reliable partner for pharma distributors, exporters, government suppliers, and PCD pharma franchise businesses across Africa, the Middle East, and Latin America (LATAM). Potassium Iodate Tablets 85mg (TAJZOSAT) – Product Overview Potassium Iodate Tablets 85mg Potassium Iodate Tablets 85mg (TAJZOSAT) are formulated as a thyroid-protective agent, primarily used in emergency medical preparedness, especially during nuclear radiation exposure scenarios. These tablets are designed to provide stable iodine to the body, which helps in saturating the thyroid gland and reducing the uptake of radioactive iodine. This significantly lowers the risk of radiation-induced thyroid damage and long-term complications such as thyroid cancer. Our formulation is developed under strict pharmaceutical controls to ensure accurate dosage, stability, and high bioavailability. Mechanism of Action – […]
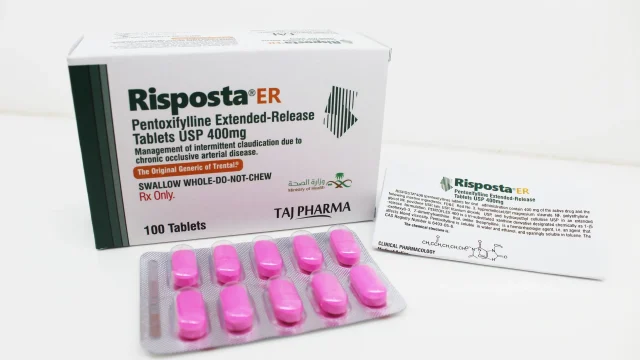
Pentoxifylline Extended-Release 400mg Tablets Manufacturer in India India has become a preferred destination for high-quality pharmaceutical manufacturing, and we proudly stand among the leading names as a Pentoxifylline Extended-Release 400mg Tablets Manufacturer in India. With advanced production facilities, strict quality systems, and a commitment to global healthcare standards, we manufacture and supply premium Pentoxifylline tablets for domestic and international markets. We specialize in delivering reliable formulations for hospitals, wholesalers, distributors, government tenders, and healthcare institutions. Our manufacturing expertise, competitive pricing, and export capabilities make us a trusted partner for clients seeking dependable Pentoxifylline Extended-Release 400mg Tablets. Pentoxifylline Extended-Release 400mg Tablets – Product Overview Pentoxifylline Extended Release 400mg Tablets Pentoxifylline Extended-Release 400mg Tablets are widely used in the management of peripheral vascular disorders. The medicine helps improve blood circulation by reducing blood viscosity and enhancing red blood cell flexibility. This supports better oxygen delivery to tissues and improves circulation in narrowed arteries. These tablets are commonly prescribed for patients experiencing poor blood flow in the legs, hands, and feet, particularly in cases of intermittent claudication associated with peripheral artery disease. Key Benefits of Pentoxifylline 400mg Tablets Supports healthy blood circulation Helps reduce leg pain caused by walking Improves oxygen supply to […]
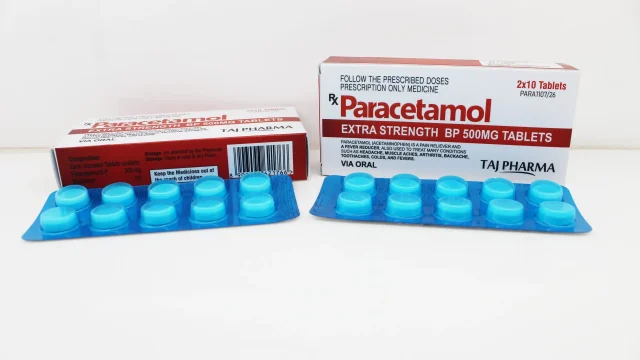
Paracetamol Tablets 500mg Manufacturer, Supplier and Exporter in India Leading Paracetamol Tablets 500mg Manufacturer in India We at Taj Medico are recognized as a trusted Paracetamol Tablets 500mg manufacturer, supplier, and exporter in India, delivering high-quality pharmaceutical formulations that meet global healthcare standards. Paracetamol (Acetaminophen) 500mg tablets are among the most widely used analgesic and antipyretic medicines, recommended for the relief of pain and fever. Our manufacturing operations are based in Vapi, Gujarat, India, and are supported by advanced production infrastructure, strict quality control systems, and experienced pharmaceutical professionals. We focus on ensuring consistent product efficacy, safety, and stability across every batch produced. Paracetamol Tablets 500mg are formulated to provide effective relief from mild to moderate pain conditions while maintaining a strong safety profile when used as directed. Paracetamol Tablets 500mg – Therapeutic Overview and Uses Paracetamol 500 mg Paracetamol 500mg is a widely prescribed medicine used for the treatment of various conditions associated with pain and fever. It works by inhibiting the production of prostaglandins in the brain, which are responsible for triggering pain and elevated body temperature. Key therapeutic uses include: Headache and migraine relief Toothache and dental pain Muscular pain and body aches Fever associated with infections […]
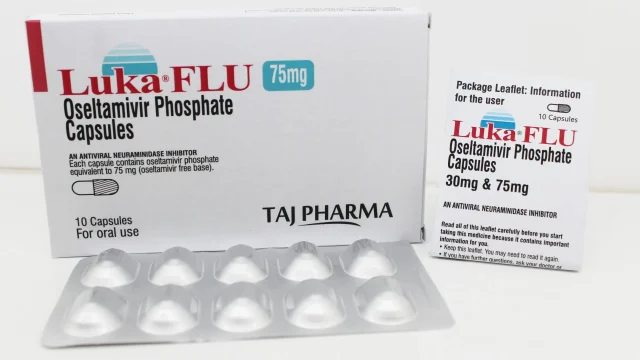
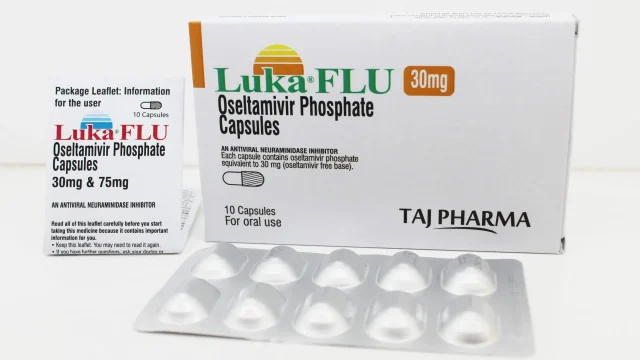
Oseltamivir Phosphate Capsules 75mg Manufacturer in India Leading Oseltamivir Phosphate Capsules 75mg Manufacturer in India We at Taj Medico India are a reputed and well-established manufacturer, exporter, and supplier of Oseltamivir Phosphate Capsules 75mg, offering high-quality antiviral formulations from our advanced manufacturing facility located in Vapi, Gujarat, India. Our product, widely recognized under the brand (Luka FLU) Oseltamivir Capsules 75mg, is developed with a strong commitment to purity, safety, and international pharmaceutical standards. Oseltamivir Phosphate Capsules are widely used in the treatment and prevention of influenza (flu), including seasonal influenza and H1N1 (Swine Flu) infections. With increasing global demand for reliable antiviral medicines, we ensure consistent supply across India, Africa, the Middle East, and Latin America (LATAM) markets. Oseltamivir Phosphate Capsules 75mg – Trusted Antiviral Formulation Oseltamivir Phosphate Capsules 75mg Oseltamivir Phosphate belongs to a class of antiviral medicines known as neuraminidase inhibitors. It works by inhibiting the activity of the neuraminidase enzyme, which is essential for the influenza virus to spread within the human body. By blocking this enzyme, the medicine helps: Reduce viral multiplication Limit infection spread in the respiratory tract Shorten flu duration and severity Support faster recovery when taken at early stages Our Oseltamivir Capsules 75mg […]
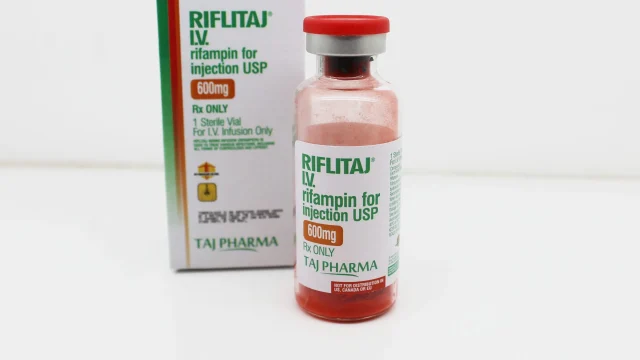
Rifampin 600mg Injection Manufacturer in India Introduction to Rifampin 600mg Injection Manufacturing in India We at Taj Medico India take pride in being a leading Rifampin 600mg Injection manufacturer in India, delivering world-class injectable formulations with a strong commitment to quality, safety, and therapeutic effectiveness. Our flagship product RIFLITAJ (Rifampin 600mg Injection) is widely recognized in domestic and international markets for its high purity standards and reliable clinical performance. Rifampin (also known as Rifampicin) is a powerful antibiotic extensively used in the treatment of tuberculosis (TB) and leprosy, and in selected severe bacterial infections where rapid antimicrobial action is required. With increasing global demand for injectable anti-tubercular drugs, we ensure uninterrupted manufacturing and supply from our advanced facility located in Vapi, Gujarat, India. What is Rifampin 600mg Injection? Rifampin 600mg Injection Rifampin 600mg Injection is a potent intravenous antibiotic formulation used primarily in hospital settings. It is especially beneficial in patients where oral therapy is not possible or immediate systemic action is required. The active ingredient, Rifampicin, belongs to the rifamycin class of antibiotics. It works by targeting bacterial RNA synthesis, thereby inhibiting the production of essential proteins required for bacterial growth and replication. We ensure that every batch of […]
Oseltamivir Phosphate Capsules 30mg Manufacturer in India Leading Oseltamivir Phosphate Capsules 30mg Manufacturer in India We are a reputed and well-established manufacturer, exporter, and supplier of Oseltamivir Phosphate Capsules 30mg in India, delivering high-quality pharmaceutical formulations under strict international standards. Our manufacturing unit is based in Vapi, Gujarat, India, and is equipped with advanced production facilities that ensure consistency, purity, and global compliance. Oseltamivir Phosphate Capsules 30mg are widely recognized for their role in the treatment and prevention of influenza (flu), including H1N1 (Swine Flu). We ensure that every capsule is manufactured under controlled environments to maintain therapeutic effectiveness and patient safety. With a strong presence in Africa, Middle East, and Latin America (LATAM), we continue to expand our footprint as a premium global supplier of antiviral medicines. High-Quality Oseltamivir Capsules 30mg for Influenza Treatment Oseltamivir Phosphate Capsules 30mg We specialize in producing Oseltamivir (Luka FLU) Capsules 30mg, formulated to help in the management of influenza virus infections. These capsules are widely prescribed for: Treatment of seasonal influenza (flu) Management of H1N1 influenza (Swine flu) Prevention of flu in high-risk exposure cases Reducing severity and duration of influenza symptoms Our Oseltamivir capsules are developed using high-grade active pharmaceutical ingredients (APIs) […]
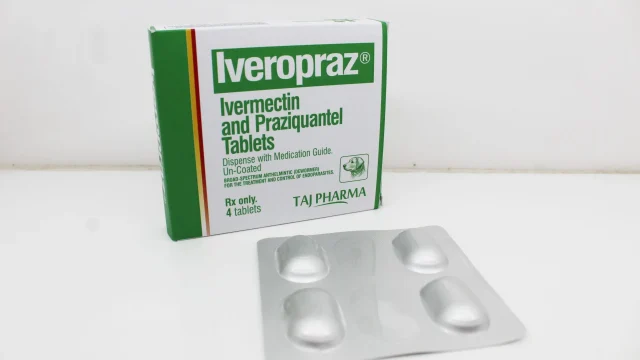
Ivermectin 2mg and Praziquantel 50mg Tablets Manufacturer in India India has become a leading hub for high-quality veterinary pharmaceutical manufacturing, offering advanced formulations for companion animal healthcare worldwide. We are recognized as a reliable Ivermectin 2mg and Praziquantel 50mg Tablets Manufacturer in India, delivering premium veterinary antiparasitic tablets designed for dogs and cats. Our manufacturing excellence, stringent quality systems, and global supply capabilities make us a preferred partner for distributors, importers, wholesalers, and veterinary businesses across multiple regions. We specialize in producing Ivermectin 2mg and Praziquantel 50mg Tablets under strict GMP-compliant standards, ensuring purity, potency, stability, and consistent therapeutic effectiveness. Our products are supplied across domestic and international markets including Africa, the Middle East, LATAM, and Asia. What Are Ivermectin 2mg and Praziquantel 50mg Tablets? Ivermectin 2mg and Praziquantel 50mg Tablets Ivermectin 2mg and Praziquantel 50mg Tablets are broad-spectrum veterinary antiparasitic tablets formulated for the prevention and treatment of internal parasitic infestations in pets. This dual-combination tablet is widely prescribed for dogs and cats to control multiple parasites in a single dose. The combination contains: Ivermectin 2mg – Effective against nematodes, heartworm larvae, and certain mites. Praziquantel 50mg – Highly effective against cestodes including tapeworm infestations. This combination helps veterinary […]
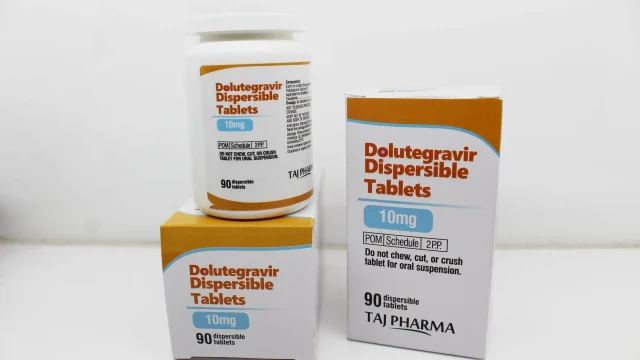
Dolutegravir Dispersible Tablets 10mg Manufacturer in India Leading Dolutegravir Dispersible Tablets 10mg Manufacturer in India We are a trusted Dolutegravir Dispersible Tablets 10mg manufacturer in India, delivering premium-quality pharmaceutical formulations that meet stringent domestic and international standards. Our commitment to innovation, regulatory compliance, and consistent product quality has positioned us among the preferred names for healthcare institutions, importers, distributors, NGOs, and pharmaceutical businesses seeking dependable HIV treatment medicines. Dolutegravir Dispersible Tablets 10mg are widely prescribed in antiretroviral therapy for pediatric and specific patient requirements. Manufactured in WHO-GMP compliant facilities, our formulations are designed to ensure dosage accuracy, stability, safety, and excellent therapeutic performance. What Are Dolutegravir Dispersible Tablets 10mg? Dolutegravir Dispersible Tablets 10mg Dolutegravir Dispersible Tablets 10mg contain Dolutegravir, an integrase strand transfer inhibitor (INSTI) used in the management of HIV-1 infection. This medicine works by preventing the virus from multiplying in the body, thereby helping control infection progression. Dispersible tablets are specially developed for convenient administration. These tablets dissolve easily in water, making them highly suitable for pediatric patients and individuals who have difficulty swallowing conventional tablets. Our manufacturing standards ensure: Accurate 10mg potency in every tablet Fast dispersion in water Uniform taste masking support Long shelf stability Moisture-protective […]
Deflazacort 6mg Tablets Manufacturer in India Deflazacort 6mg Tablets Leading Manufacturer in India We are recognized as a reliable Deflazacort 6mg Tablets manufacturer in India, delivering premium quality pharmaceutical formulations for domestic and international markets. With advanced manufacturing capabilities, strict quality systems, and customer-focused service, we supply Deflazacort tablets across PAN India and to global destinations including Africa, the Middle East, and LATAM countries. Our manufacturing unit in Vapi, Gujarat, India follows GMP standards and modern pharmaceutical protocols to ensure purity, consistency, safety, and regulatory compliance. We manufacture Deflazacort 6mg tablets under the brand DMDTAJ, developed to meet rising healthcare demand for corticosteroid therapy. What is Deflazacort 6mg Tablet? Deflazacort 6mg Tablets Deflazacort 6mg Tablet is a corticosteroid medicine widely used in the treatment of Duchenne Muscular Dystrophy (DMD) in patients aged 5 years and older. It is also prescribed for inflammatory disorders, autoimmune conditions, allergic reactions, and other medical indications where anti-inflammatory action is required. This medicine helps reduce inflammation, suppress harmful immune responses, preserve muscle strength, and slow muscle weakness progression in DMD patients. Deflazacort is considered a preferred corticosteroid option due to its favorable tolerability profile when prescribed correctly. Deflazacort 6mg Tablets Uses We manufacture Deflazacort 6mg […]
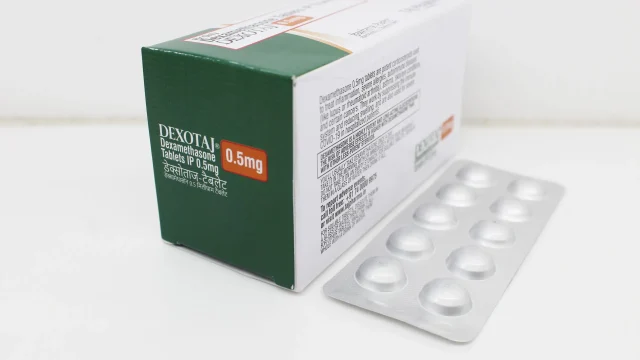
Dexamethasone Tablets 0.5mg Manufacturer in India Leading Dexamethasone Tablets 0.5mg Manufacturer in India We are proud to be recognized as a trusted Dexamethasone Tablets 0.5mg manufacturer in India, delivering superior pharmaceutical products to domestic and international markets. Our Dexamethasone Tablets 0.5mg are manufactured under strict WHO-GMP quality standards using advanced production technology and premium-grade raw materials. We focus on purity, safety, accurate dosage, and consistent therapeutic performance, making us a preferred source for distributors, hospitals, pharmacies, and global buyers. Our flagship brand Dexotaj has earned strong market trust for quality packaging, dependable results, and competitive pricing. We supply across PAN India and export to Africa, the Middle East, LATAM, and many other international regions. What is Dexamethasone Tablets 0.5mg? Dexamethasone Tablets Dexamethasone Tablets 0.5mg belong to a group of medicines known as corticosteroids. These tablets are widely used in the treatment of inflammatory disorders, autoimmune diseases, allergic reactions, respiratory conditions, skin disorders, hormonal conditions, bowel disorders, and supportive cancer care. They help reduce swelling, redness, irritation, and immune overactivity when prescribed by healthcare professionals. Our formulation is manufactured with strict pharmaceutical controls to ensure stability, efficacy, and safety. Why Choose Us as Dexamethasone Tablets Manufacturer & Supplier We maintain a […]
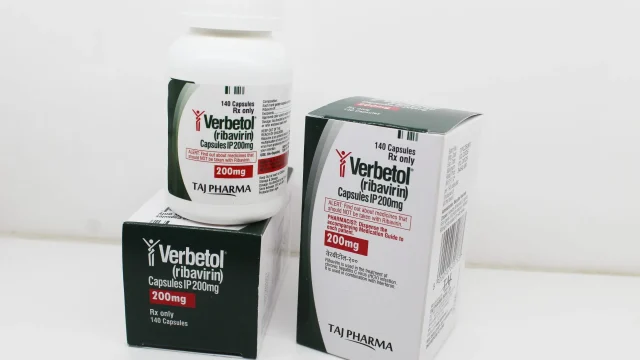
Ribavirin Capsules 200mg Manufacturer Supplier in India 💊 Leading Ribavirin Capsules 200mg Manufacturer in India We are a reputed Ribavirin Capsules 200mg manufacturer in India, delivering high-quality antiviral pharmaceutical products with complete assurance of purity, safety, and effectiveness. Our modern manufacturing facilities follow stringent WHO-GMP quality systems to ensure that every batch of Ribavirin 200mg Capsules meets national and international standards. We proudly serve pharmaceutical distributors, hospitals, healthcare institutions, wholesalers, exporters, and franchise partners across India and global markets. Our company has earned a strong position among trusted Ribavirin Capsules 200mg suppliers in India by offering reliable stock availability, professional service, timely dispatch, and competitive pricing. 🏭 Premium Quality Ribavirin Capsules 200mg Manufacturing Ribavirin Capsules 200mg Our production infrastructure is equipped with advanced machinery, cleanroom technology, quality testing laboratories, and experienced pharmaceutical professionals. Each capsule is manufactured with carefully selected raw materials to maintain consistent potency and stability. Our Manufacturing Strengths ✔ WHO-GMP compliant production units ✔ Advanced capsule filling technology ✔ Strict in-process quality checks ✔ Hygienic packaging standards ✔ Large volume manufacturing capacity ✔ Customized labeling and private branding This makes us one of the most dependable Ribavirin Capsules 200mg manufacturers PAN India. 📌 What is Ribavirin Capsules […]
Could a protein grown in lettuce help heal broken bones faster {Taj Pharma Regulatory Affairs}
12 -Foods to boost brain function {Taj Medico}
[Liver cancer] deaths climb by around 50% in the last decade {Taj Pharmaceuticals Survey}
Blood Tests: Using Blood to detect Cancer Early {Cancer is word; not a sentence} [Taj Pharmaceuticals Mumbai]
WHO releases guideline on digital health interventions {Taj Pharmaceuticals} [Patient Care]





![[Liver cancer] deaths climb by around 50% in the last decade {Taj Pharmaceuticals Survey}](https://tajmedico.com/wp-content/uploads/cache/images/Liver-cancer-deaths-climb-by-around-50-in-the-last-decade-Taj-Pharmaceuticals-Survey-2-scaled/Liver-cancer-deaths-climb-by-around-50-in-the-last-decade-Taj-Pharmaceuticals-Survey-2-scaled-860068172.jpg)
![Blood Tests: Using Blood to detect Cancer Early {Cancer is word; not a sentence} [Taj Pharmaceuticals Mumbai]](https://tajmedico.com/wp-content/uploads/cache/images/Blood-Tests-Using-Blood-to-detect-Cancer-Early-Cancer-is-word-not-a-sentence-Taj-Pharmaceuticals-Mumbai/Blood-Tests-Using-Blood-to-detect-Cancer-Early-Cancer-is-word-not-a-sentence-Taj-Pharmaceuticals-Mumbai-2746981357.jpg)
![WHO releases guideline on digital health interventions {Taj Pharmaceuticals} [Patient Care]](https://tajmedico.com/wp-content/uploads/cache/images/WHO-releases-guideline-on-digital-health-interventions-Taj-Pharmaceuticals-Patient-Care-by-Taj-Pharma-Group-Taj-Medico/WHO-releases-guideline-on-digital-health-interventions-Taj-Pharmaceuticals-Patient-Care-by-Taj-Pharma-Group-Taj-Medico-1592706783.jpg)





